Biocryst: Orladeyo
Launching into a crowded rare market
INSIGHT
Hereditary angioedema (HAE) puts a heavy weight on patients’ shoulders. Whether they are experiencing a debilitating attack, fearing their next attack, or struggling with the burden of treatment, patients lose precious time and attention to HAE. HCPs, meanwhile, have grown accustomed to prescribing a range of injectable and intravenous HAE treatments whose administration and storage requirements only add to the patient burden.
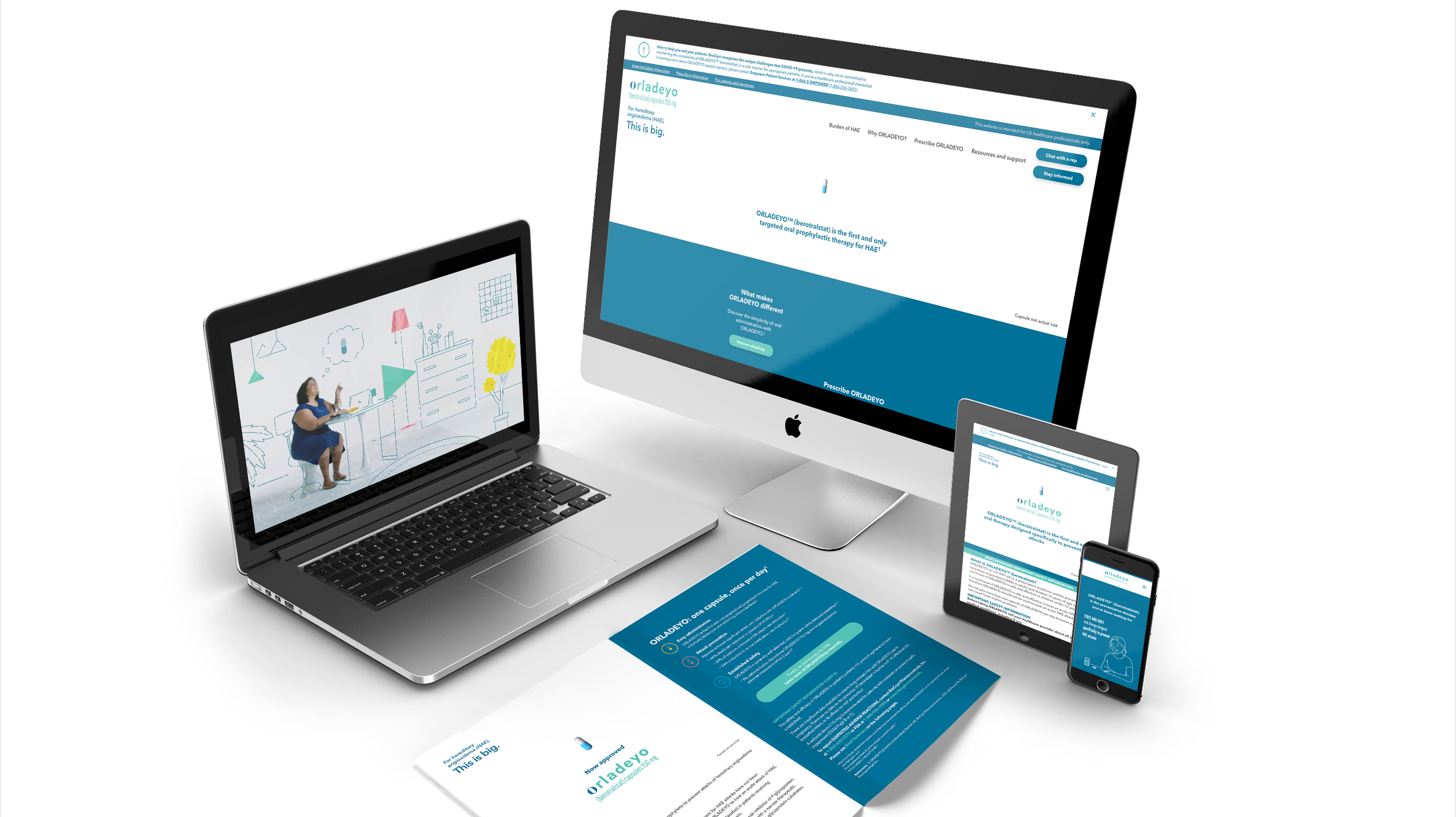
Enter Orladeyo, the first and only oral prophylactic therapy designed to help minimize the
time spent treating HAE. This could help to lighten patients’ burden, but we faced a crowded market conditioned to maintain the status quo, and a hesitant patient population.
IDEA
We knew that to launch Orladeyo successfully in December 2020, we had to cut through the noise: first by capturing prelaunch attention early and often, and then by developing a creative campaign to stand out amid market clutter. To do this, we utilized something unexpected:
white space. We allowed the single Orladeyo capsule to speak for itself in a minimalist campaign that invited people to come closer. To listen. “This is big,” we whispered in the headline. Would they hear us?
IMPACT
They did. Within 3 months of launch, the Orladeyo patient website had 15,000 visitors, 525
discussion-guide downloads, and more than 200 sign-ups. The HCP website had 7,000 visitors,
540 start-form downloads, and more than 40 sign-ups. BioCryst reported $10.9 million in sales in the first 3 months and $122 million for the first year, blowing expectations out of the water. And the success continued into year 2. As Jon Stonehouse, President and CEO of BioCryst, said: “In our second year on the market, we more than doubled our first-year ORLADEYO sales. This continues to be an exceptional launch of an oral rare disease drug, and we expect this success to continue creating real value for patients and for shareholders this year and for many years to come.”